Introduction
Pneumomediastinum is often observed in the setting of oesophageal rupture (Boerhaave’s syndrome), trauma resulting in tracheo-bronchial rupture, acute severe asthma (ASA) and mediastinitis due to gas-producing organisms. Spontaneous pneumomediastinum (SPM) is defined as free air in the mediastinum caused by nontraumatic and noniatrogenic mechanisms in the absence of pre-existing parenchymal or obstructive lung disease. SPM was first described in a postpartum female following vaginal delivery.1 SPM is an uncommon entity with an incidence of 0.001–0.01%.2 H1N1 infection is rarely associated with SPM. It is crucial to recognise this condition so that the patient is monitored in the intensive care unit (ICU). Though SPM is broadly regarded a benign entity, one should be cautious about its evolution into pneumothorax, tension pneumomediastinum and extensive subcutaneous emphysema (SCE) in rare cases, which can cause cardiorespiratory compromise culminating in death. In this paper, we elucidate the pathophysiology of SPM and its management.
Case presentation
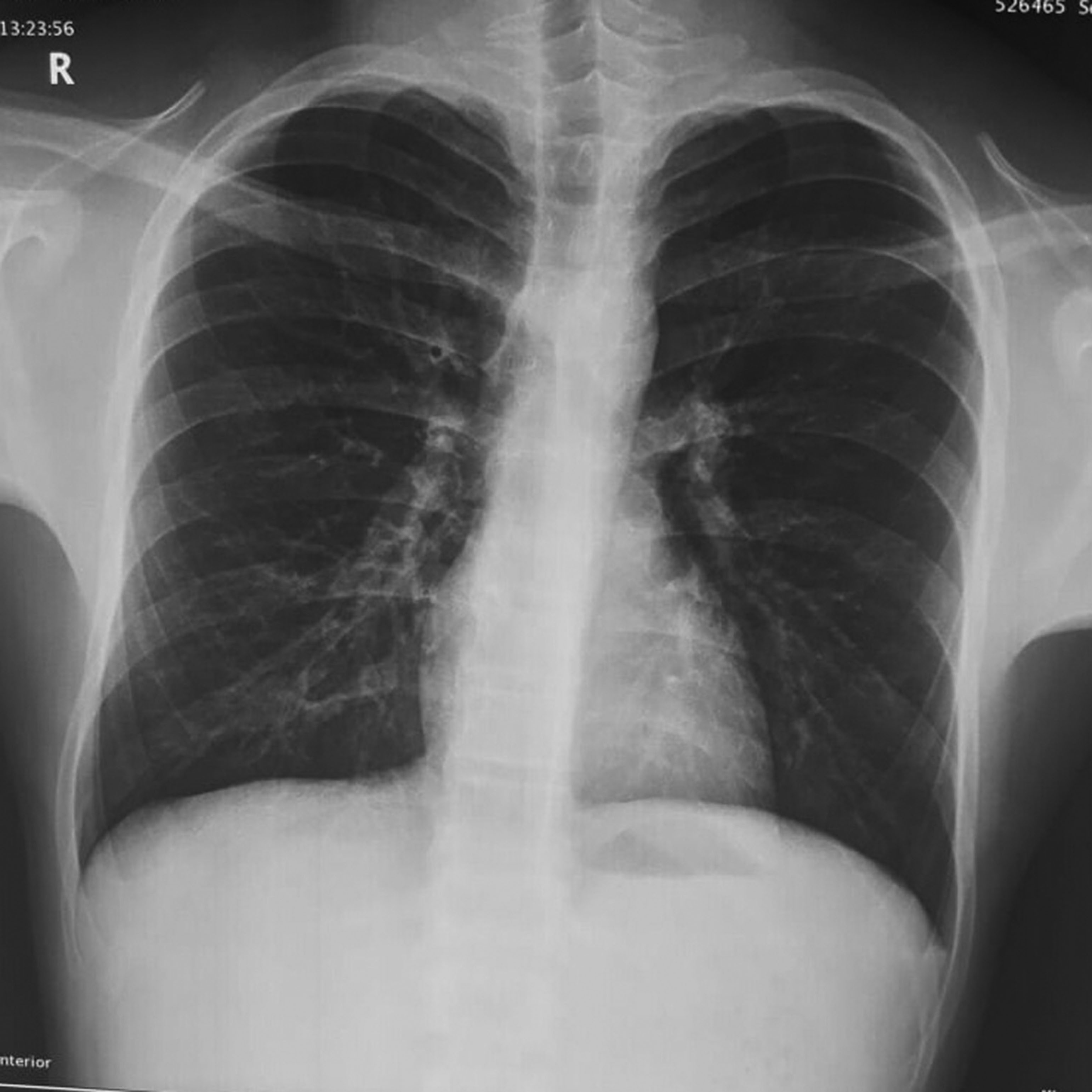
A 17-year-old male, presented to the emergency department with history of fever, dry cough, throat pain for 1 day and acute onset shortness of breath associated with wheeze for 2 hours. He was conscious, had tachypnoea (respiratory rate: 33/min), tachycardia (pulse rate: 140/min) and his oxygen saturation (SpO2) was 88%. He had swelling with palpable crepitus over left infraclavicular, axillary areas and over the anterior aspect of neck on the left side suggestive of SCE. Ronchi were heard all over the chest. He came with a chest roentgenogram performed that morning that appeared normal. Arterial blood gas analysis showed features of type 1 respiratory failure (pH: 7.35; PaO2: 60 mm Hg; PaCO2: 32 mm Hg; bicarbonate: 25.3 mmol/l). Complete blood count, renal and liver function tests were normal. Chest roentgenogram confirmed SCE (Figure 1). Reports of SPM associated with SCE in H1N1 infection prompted us to obtain CT thorax, as the latter cannot be diagnosed on chest roentgenogram. CT showed patchy ground-glass opacities in the bilateral lung fields suggesting atypical pneumonia. Pneumomediastinum with air tracking into the central compartment of the neck and extensive SCE in the neck, left chest wall and axilla were also noted on CT images (Figure 2). Throat swab specimen was positive for H1N1 influenza virus by polymerase chain reaction. He was managed initially in ICU with oral oseltamivir, parenteral and inhalational steroids, inhalational short-acting β agonist and oxygen therapy. He improved clinically by the third day. SCE subsided and he was discharged on the fourth day. He was advised absolute bed rest at home. On follow up after 7 days, chest X-ray showed complete resolution of SCE (Figure 3).
Figure 1 Chest X-ray (anteroposterior view) showing subcutaneous emphysema in the chest wall and neck (arrows)
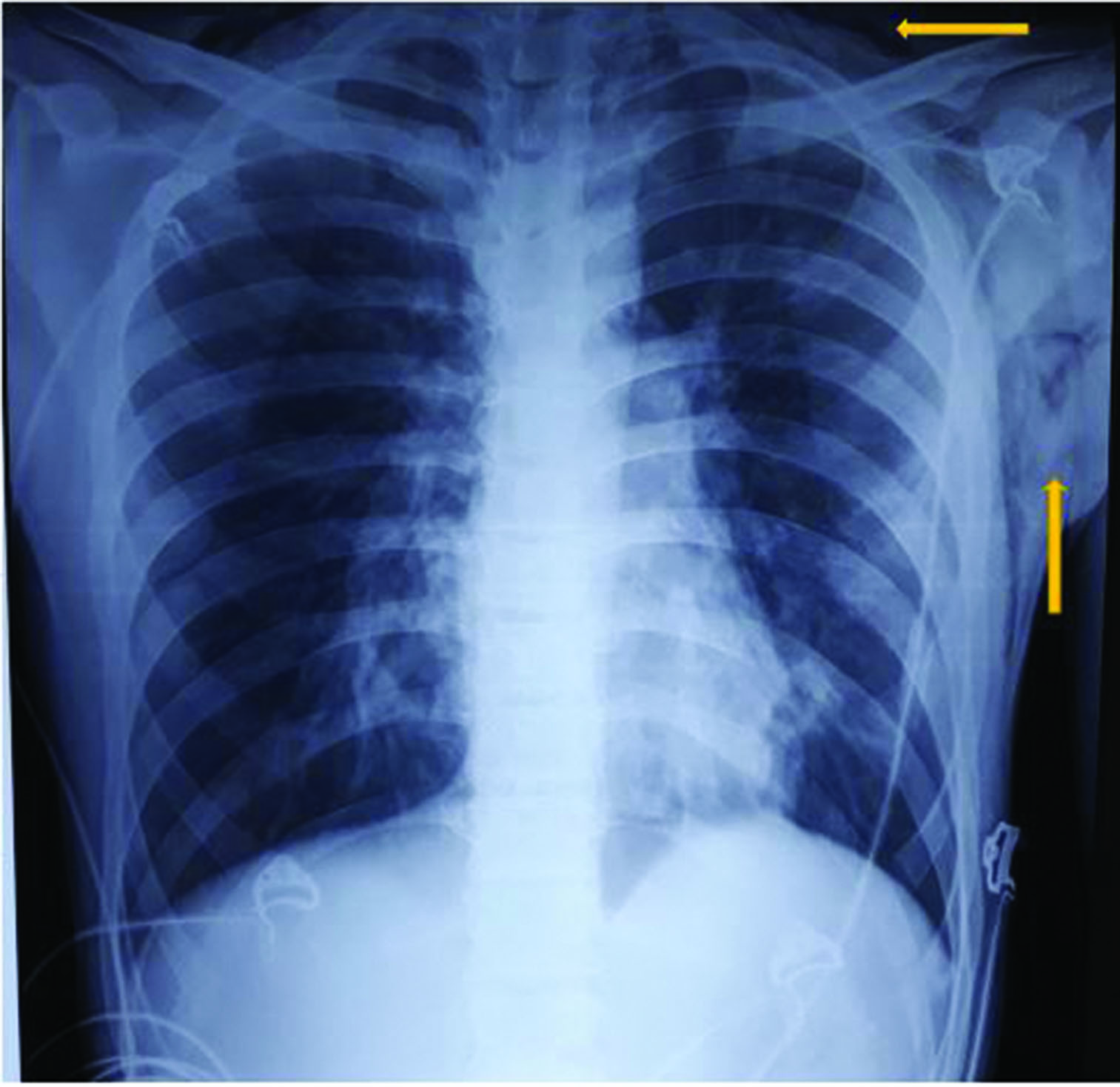
Figure 2 CT chest (axial plane) showing extensive subcutaneous emphysema and pneumomediastinum (yellow arrows); patchy ground-glass opacities in bilateral lung fields (red arrow)
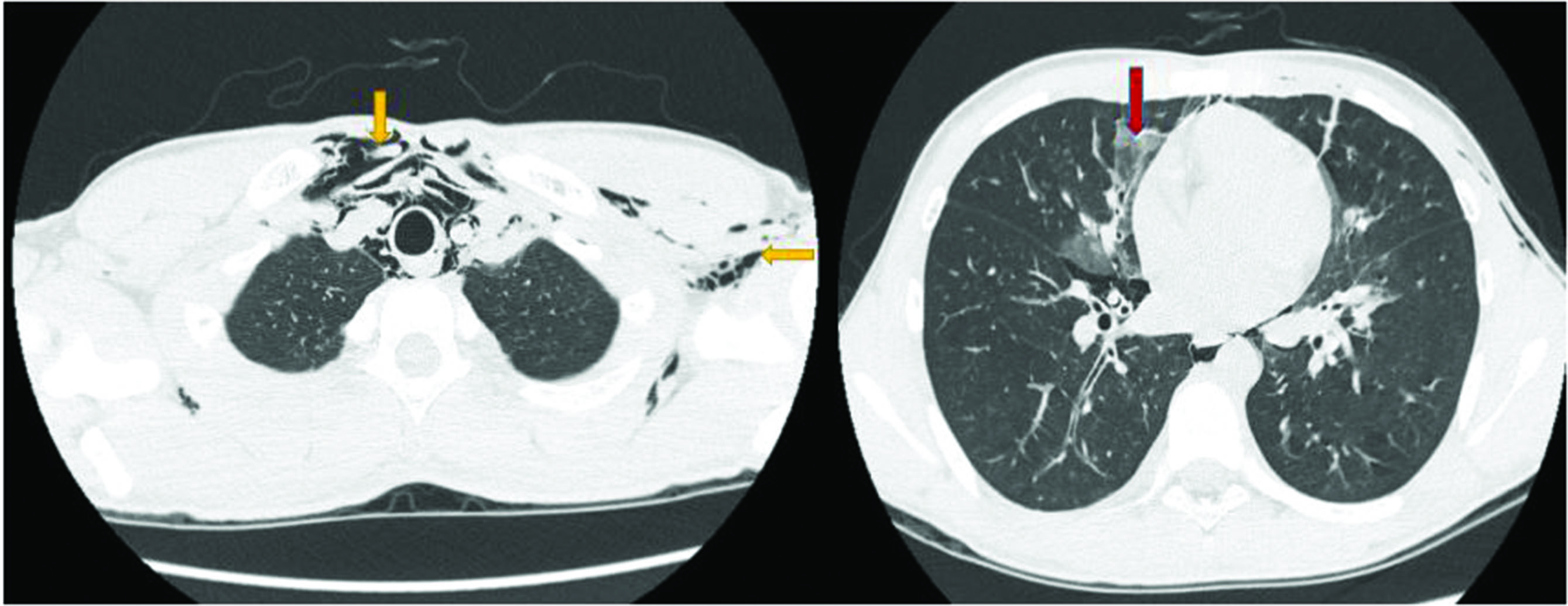
Figure 3 Repeat chest X-ray (posteroanterior view) after 1 week showing complete resolution of subcutaneous emphysema
Discussion
The description of SPM was first given by Louis Bourgeois in the seventeenth century. He conceptualised that Valsalva performed during vaginal delivery could be responsible for SPM.1 Aetiology of SPM includes bronchial asthma, straining during exercise, intense cough, retching, diabetic ketoacidosis and inhalational drugs (cocaine, marijuana), among others. Other rare associations with yoga, gastroesophageal reflux, anorexia nervosa, Hodgkin’s lymphoma after chest irradiation and infections, such as H1N1, are described in recent case reports. In a study by Chapdelaine et al.,3 the most common aetiology of SPM was bronchospasm (49%) followed by respiratory infections (21%) and unknown aetiology (15%). In another study, unknown aetiology (34%), drug abuse (14%), exercise (14%), intense cough (10%), ASA (9%), vomiting (7%), respiratory infections (5%), cocaine (2%), cannabis (2%), amphetamine (1%) and prolonged shouting (1.5%), among others, have been reported as the triggering factors.4 Our patient denied history of any of these factors and, hence, we attributed SPM in this case to H1N1 infection.
Macklin and Macklin5 explained the pathophysiology of SPM based on animal models. Excessive distension of alveoli results in their rupture into the vascular sheath, which is in anatomic continuity with mediastinal space and subcutaneous facial planes. This results in escape of air into mediastinal space and facial planes resulting in SPM and SCE. Air can also enter the pleural cavity resulting in pneumothorax.6 Alveolar rupture occurs by two mechanisms that create a pressure gradient between intra-alveolar and interstitial spaces – increase in intra-alveolar pressure or decrease in peri-alveolar interstitial space pressure. The former phenomenon is seen in Valsalva manoeuvre, ASA, violent cough and obstruction to expiratory flow in a ventilatory circuit. The latter phenomenon is seen in strenuous exercise, marijuana smoking, trekking, scuba diving and intense respiratory effort.1 Young patients like ours are at high risk for SPM and SCE owing to lax mediastinal tissues. Elderly patients have fibrosed tissues making them taut that prevents escape of air from the airway. Though SPM is considered an uncommon condition, the recent rise in the number of cases can be ascribed to widespread availability of diagnostic modalities, such as chest CT, and an increase in epidemic respiratory infections that mainly affect the young population.
H1N1 infection has not been described as a trigger in the studies on SPM so far;4,7 however, a few cases have been reported in the literature. The Novel Swine-Origin Influenza A (H1N1) Virus Investigation Team, reported SPM complicating H1N1 infection for the first time in one case.8 A few such cases have been reported subsequently and the hallmark feature was all these patients were aged >10 years.9–11 The mechanism of SPM in the context of H1N1 infection can be explained by either alveolar damage associated with the infection or intense cough, or both.10,11
The most common presenting feature of SPM is chest pain, but chest pain was not prominent in our patient. Other features include dyspnoea, cough and dysphagia, among others. The classical examination finding in SPM is Hamman’s sign. It is described as a crunching or clicking sound heard on auscultation over the apex and along the left heart border synchronous with the heartbeat. It occurs owing to heart beating against tissues filled with air. Our patient lacked this finding. SCE and SPM coexist in 70% of the patients.12 In a study by Mondello et al.,13 chest pain was described as the most common symptom (100%), followed by neck pain (44%), dyspnoea (88%), cough (77%), nasal voice (66%) and dysphagia (22%). Thoracic pain (61%), dyspnoea (41%), subcutaneous thoracic emphysema (40.3%), persistent cough (20%), cervical pain (16.5%), subcutaneous cervical emphysema (14.5%), dysphagia (14%), Hamman’s sign (13.8%), cervical oedema (8.8%), dysphonia (4.8%), odynophagia (3.8%), rhinolalia (3.3%), fever (3.2%), thoracic pain plus dyspnoea (2.7%), dizziness (2.5%), crepitus sensation (2%), back pain (1.7%), generalised weakness (1.7%), stridor (1.5%), anxiety (0.7%), abdominal pain (0.7%), tachycardia (0.7%), pulsus paradoxus (0.3%), pharyngeal enlargement (0.2%) and massive SCE (0.2%) have been described as the clinical features of pneumomediastinum in a study by Dajer-Fadel et al.4
SPM can be seen as air tracking along the left cardiac border on a chest X-ray. However, it might be arduous to recognise SPM if the findings are subtle. At times it may also be difficult to diagnose a coexisting trivial pneumothorax. Therefore, once SCE is identified, it is important to obtain a CT chest in the setting of H1N1 influenza.
Nonetheless, SPM is usually considered a benign entity. Rarely, it might lead to complications, such as pneumothorax and tension pneumomediastinum, that compromise cardiac output. ‘Air block’, a phenomenon of pressure effect on the airway caused by pneumomediastinum, can produce further respiratory distress.1 Tension pneumothorax, worsening of presenting symptoms and recurrent SPM were described as the complications of SPM in a study by Dajer-Fadel et al.4 Therefore, identifying SPM and keeping close watch on the patient both during admission and on follow up are necessary. Management should be focussed on treating the primary disease. Specific therapy for SPM includes high-flow oxygen inhalation, absolute bed rest and close monitoring. The outcome is generally positive as in our patient.
Conclusion
This case report brought an unusual complication of H1N1 influenza to the attention of the medical fraternity. SPM is mostly seen in young individuals. Chest pain, neck pain, SCE and Hamman’s sign on auscultation in the presence of appropriate inciting factors should prompt a clinician to consider SPM. It is rarely fatal but requires close monitoring. Treating the inciting factor generally resolves SPM. In young patients, asthma, drug abuse and history of strenuous exercise should be excluded first, as these are more common inciting factors than H1N1 infection. Nevertheless, the treatment principles remain the same.
References
1 Maunder RJ, Pierson DJ, Hudson LD. Subcutaneous and mediastinal emphysema: pathophysiology, diagnosis and management. Arch Intern Med 1984; 144: 1447–53.
2 Takada K, Matsumoto S, Hiramatsu T et al. Spontaneous pneumomediastinum: an algorithm for diagnosis and management. Ther Adv Respir Dis 2009; 3: 301–7.
3 Chapdelaine J, Beaunoyer M, Daigneault P et al. Spontaneous pneumomediastinum: are we overinvestigating? J Pediatr Surg 2004; 39: 681–4.
4 Dajer-Fadel WL, Argüero-Sánchez R, Ibarra-Pérez C et al. Systematic review of spontaneous pneumomediastinum: a survey of 22 years’ data. Asian Cardiovasc Thorac Ann 2014; 22: 997–1002.
5 Macklin MT, Macklin CC. Malignant interstitial emphysema of the lungs and mediastinum as an important occult complication in many respiratory diseases and other conditions: an interpretation of the clinical literature in the light of laboratory experiment. Medicine 1944; 23: 281–352.
6 Meyers MA. Radiological features of the spread and localization of extraperitoneal gas and their relationship to its source. Diagn Radiol 1974; 111: 17–26.
7 Dionísio P, Martins L, Moreira S et al. Spontaneous pneumomediastinum: experience in 18 patients during the last 12 years. J Bras Pneumol 2017; 43: 101–5.
8 Dawood FS, Jain S, Finelli L et al.; Novel Swine-Origin Influenza A (H1N1) Virus Investigation Team. Emergence of a novel swine-origin influenza A (H1N1) virus in humans. N Engl J Med 2009; 360: 2605–15.
9 Hasegawa M, Hashimoto K, Morozumi M et al. Spontaneous pneumomediastinum complicating pneumonia in children infected with 2009 pandemic influenza A (H1N1) virus. Clin Microbiol Infect 2010; 16: 195–9.
10 Udupa S, Hameed T, Kovesi T. Pneumomediastinum and subcutaneous emphysema associated with pandemic (H1N1) influenza in three children. CMAJ 2011; 183: 202–12.
11 Patra PK, Nayak US, Sushma TS. Spontaneous pneumomediastinum in H1N1 infection. Indian Pediatr 2011; 48: 976–7.
12 Kouritas VK, Papagiannopoulos K, Lazaridis G et al. Pneumomediastinum. J Thorac Dis 2015; 7: S44–9.
13 Mondello B, Pavia R, Ruggeri P et al. Spontaneous pneumomediastinum: experience in 18 adult patients. Lung 2007; 185: 9–14.
