Introduction
Ammonia is naturally produced as part of the metabolic process, including bacterial hydrolysis of urea in the intestine, the purine nucleotide cycle and amino acid transamination in skeletal muscle, as well as metabolic processes occurring primarily in the kidneys and the liver. It is usually eliminated by the liver as part of the urea cycle.
Failure of normal metabolism of ammonia can result in hyperammonaemia, defined in adults as an ammonia level >50 g/dl. The clinical spectrum of presentation varies from lethargy, nausea and vomiting, headache, ataxia, epilepsy to encephalopathy.1
Hyperammonaemia can be categorised as either primary or secondary. Primary causes tend to present at an earlier age and are due to enzymopathies affecting either the urea cycle or fatty acid oxidation.2 Secondary hyperammonaemia may be due to an underlying hepatic pathology or nonhepatic causes, including drug induced (valproate,3 salicylate4 and 5-fluorouracil5), post roux-en-y gastric bypass or the use of total parenteral nutrition.
Case presentation
A 44-year-old female with a background of cerebral palsy, learning disability and worsening epileptic control presented with lethargy, reduced oral intake and increasing shortness of breath. On admission she suffered a prolonged tonic–clonic seizure, which was terminated by lorazepam. Following this, her Glasgow Coma Score (GCS) declined to 6/15. A CT head scan showed no acute pathology. Arterial blood gas showed moderate respiratory acidaemia. Routine blood samples were retrieved from the patient (Table 1).
Table 1 Blood results from admission until diagnosis
| | | |
Albumin (g/l) | 31* | 30* | 27* |
Alkaline phosphatase (IU/l) | 244* | 521* | 584* |
Alanine transaminase (IU/l) | 63 | 197* | 223* |
Total bilirubin (µmol/l) | 4 | 4 | 4 |
Prothrombin time (s) | 12.3 | 11.7 | – |
Activated partial thromboplastin time (s) | 40.1* | 37.7* | – |
Sodium (mEq/l) | 141 | 140 | 141 |
Potassium (mEq/l) | 4.6 | 5.1 | 5.1 |
Urea (mmol/l) | 3.6 | - | 4.1 |
Creatinine (mmol/l) | 32 | 27 | 27 |
She was transferred to the critical care unit for respiratory support. Despite the initiation of continuous positive airway pressure and correction of her respiratory acidosis, her conscious level failed to improve.
Empirical treatment for encephalitis was initiated while awaiting lumbar puncture, which was not performed owing to technical difficulties. Over the next 4 days her liver function tests showed a gradual derangement. An ultrasound of the liver showed no abnormalities.
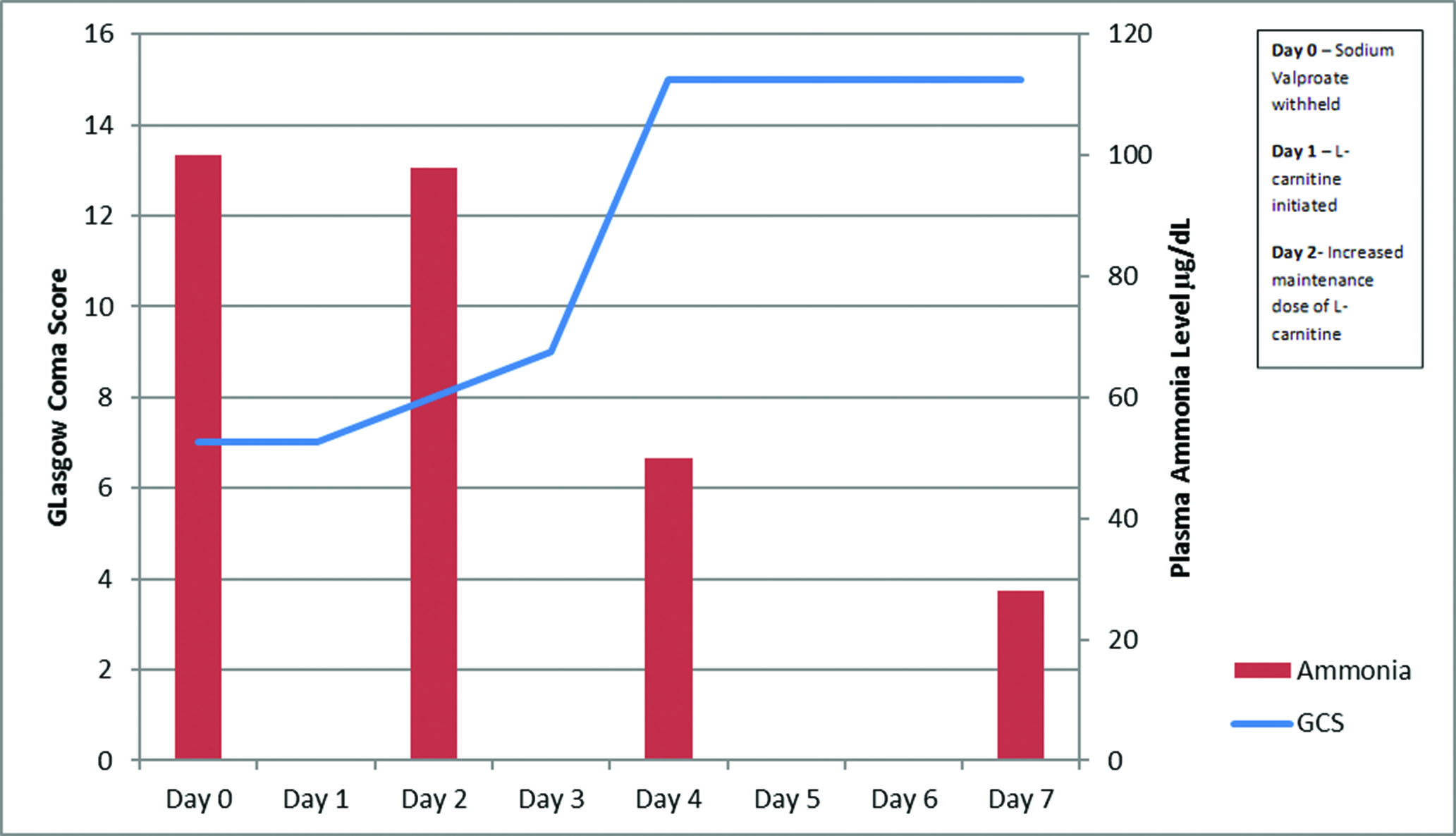
There was no improvement in conscious levels 96 hours following admission and so plasma ammonia levels were performed, and found to be raised at 100 µg/dl (normal range: 16–40 µg/dl). A suspected diagnosis of valproate-induced hyperammonaemic encephalopathy was made, sodium valproate was withheld and she was started on lactulose and levetiracetam. Unfortunately, no valproate level was performed to confirm the diagnosis.
There was no substantial change in GCS, which remained at 7/15, over the following 24 hours despite withholding the valproate. Therefore, intravenous L-carnitine was initiated with bolus dose of 6 g, then 6 hours later followed by three doses of 1 g 4 hourly. The next day ammonia levels remained unchanged at 98 µg/dl, with little improvement in her cognitive level. The maintenance dose was, therefore, increased to 3 g every 6 hours. Ammonia levels were taken 48 hours following this. They showed a reduction in plasma ammonia to 50 µg/dl and return of cognitive level to her baseline (Figure 1).
Figure 1 Graph displaying ammonia levels and Glasgow Coma Score (GCS) throughout admission, following diagnosis of valproate-induced hyperammonaemia and withdrawal of sodium vaproate, indicating timing of interventions.
Discussion
At 2 months following this admission, the patient’s carers noted that a decreased cognitive level had been apparent since the initiation of sodium valproate, 6 months prior to the admission with an increase in dose 3 months after initiation. The difference in cognitive level had completely reversed following termination of the medication. This clinical correlation supports the diagnosis of valproate-induced hyperammonaemic encephalopathy, with further evaluation of partial enzymopathies to be considered.
Acute deterioration in mental status is a common presentation to hospital. In cases where a common organic cause of this decline in cognitive status has not been identified, consideration of nonhepatic causes of hyperammonaemia should be considered.
Sodium valproate is currently the first-line antiepileptic drug for generalised seizures except in women of child-bearing age, owing to potential teratogenic effects. This exception has only been the case since April 2018.6
Ammonia levels are rarely performed, possibly owing to lack of awareness, along with the difficulty of handling the specimen. Therefore, analysis is delayed, resulting in inaccurate results.7 It has been suggested that carnitine supplementation would be beneficial in cases of severe valproate-induced hyperammonaemia, which is determined by the presence of any of the following: coma, hepatoxicity, valproic acid serum concentration >450 mcg/ml (>3,120 µmol/l) or hyperammonaemic encephalopathy. Intravenous carnitine therapy has been associated with a marked increase in survival, in comparison to oral therapy.8 The treatment of 50–100 mg/kg was based on a series of case studies and a literature review, as there are currently no randomised control trials.9
The recovery may have been due to the removal of sodium valproate. L-carnitine was initiated after 1–3 half-lives, based on the reported 7–13 hour half-life of sodium valproate, and, therefore, had not met the widely accepted criteria of 4 half-lives before being considered a negligible concentration within the body.10 Following the infusion of L-carnitine, there was a swift decrease in ammonia levels and GCS, which has been reflected in other reports.11
Conclusion
The present case highlights that hyperammonaemia should be considered as a differential in those with unexplained acutely decreased mental status. As sodium valproate is a commonly used drug, clinicians should be aware of hyperammonaemia as a potential complication.
References
1 Chawla J. Hyperammonemia Clinical Presentation. 2017. https://emedicine.medscape.com/article/1174503-clinical#b1 (accessed 23/03/28).
2 Weng TI, Shih FF, Chen WJ. Unusual causes of hyperammonemia in the ED. Am J Emerg Med 2004; 22: 105–7.
3 Tsai MF, Chen CY. Valproate-induced hyperammonemic encephalopathy treated by hemodialysis. Ren Fail 2008; 30: 822–4.
4 Makela AL, Lang H, Korpela P. Toxic encephalopathy with hyperammonaemia during high-dose salicylate therapy. Acta Neurol Scand 1980; 61: 146–56.
5 Nott L, Price TJ, Pittman K et al. Hyperammonemia encephalopathy: an important cause of neurological deterioration following chemotherapy. Leuk Lymphoma 2007; 48: 1702–11.
6 NICE. Epilepsies: diagnosis and management. 1.8 Management. https://www.nice.org.uk/guidance/cg137/chapter/1-Guidance#management-2 (accessed 23/06/2019).
7 Upadhyay R, Bleck TP, Busl KM. Hyperammonemia: what urea-lly need to know: case report of severe noncirrhotic hyperammonemic encephalopathy and review of the literature. Case Rep Med 2016; 2016: 8512721.
8 Bohan TP, Helton E, McDonald I et al. Effect of L-carnitine treatment for valproate-induced hepatotoxicity. Neurology 2001; 56: 1405–9.
9 Perrott J, Murphy NG, Zed PJ. L-carnitine for acute valproic acid overdose: a systematic review of published cases. Ann Pharmacother 2010; 44: 1287–93.
10 Medscape. Valproic acid (Rx). https://reference.medscape.com/drug/depakene-stavzor-valproic-acid-343024#showall (accessed 23/06/2019).
11 Cattaneo CI, Ressico F, Valsesia R et al. Sudden valproate-induced hyperammonemia managed with L-carnitine in a medically healthy bipolar patient: essential review of the literature and case report. Medicine (Baltimore) 2017; 96: e8117.
