A 20-year-old male with severe learning disability was referred to the neurology clinic following new onset of tonic–clonic seizures. The cause of his learning disability was uncertain but a peroxisomal disorder, possibly infantile Refsum’s disease, had been suspected because of early onset blindness although metabolic investigations in childhood had been nondiagnostic. His behavioural and linguistic issues had limited further investigations.
Seizures initially settled on sodium valproate but this medication was associated with significant weight gain. Switching to lamotrigine resulted in only partial seizure control. Owing to the uncertainty about the underlying diagnosis, and because seizures do not appear to be a typical clinical feature in infantile Refsum’s disease,1,2 further investigation was indicated.
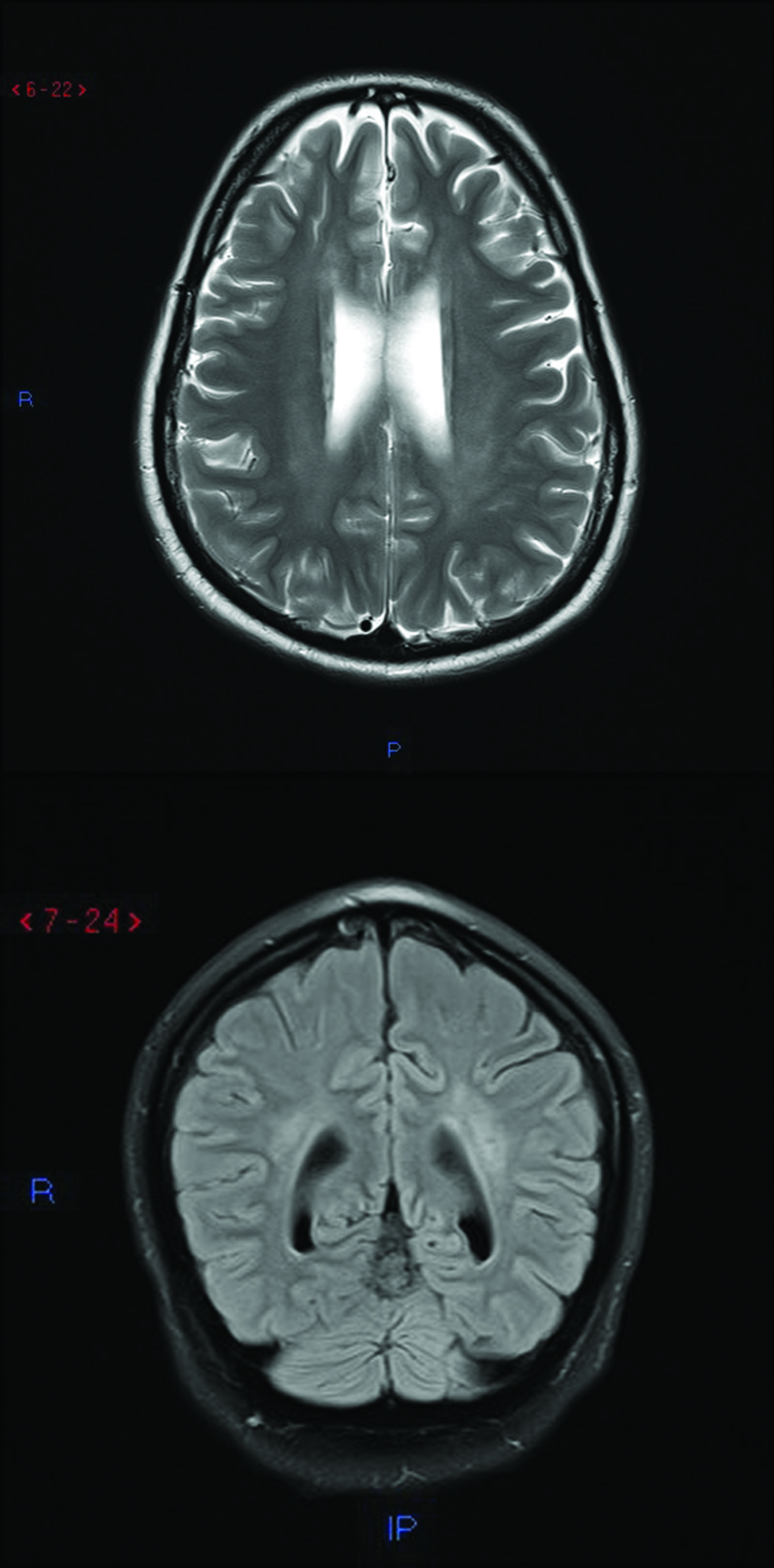
MR brain imaging (performed under general anaesthesia with parental consent) showed diffuse confluent signal hyperintensity in the white matter of the cerebral hemispheres with sparing of the subcortical U fibres (Figure 1), with additional involvement of the central cerebellum. Diffusion-weighted imaging showed no restriction. These changes were thought to be compatible with a peroxisomal disorder. Analysis of the various PEX genes showed two mutations in the PEX1 gene located on chromosome 7q21, namely c.2528G>A, p.Gly843Asp, and c.2916delA, p.Gly973AlafsTer16, confirming the diagnosis of infantile Refsum’s disease.
Figure 1 MR brain imaging: axial T2-weighted (top) and sagittal fluid-attenuated inversion recovery (bottom) images showing diffuse confluent signal hyperintensity in cerebral white matter with sparing of the subcortical U fibres
Infantile Refsum’s disease is a peroxisome biogenesis disorder that falls within the Zellweger disorder spectrum, sharing similar clinical and biochemical features but the clinical picture is less severe such that some patients survive to adulthood. Peroxisome biogenesis disorders are usually caused by biallelic mutations in the 13 PEX genes.1 The p.Gly843Asp mutation (old nomenclature: G843D) has been found in cases of infantile Refsum’s disease,3 and accounted for over 80% of all abnormal PEX1 alleles in a study of 168 Zellweger spectrum patients; the deletion c.2916delA is also described.4
The MR imaging changes seen in our patient were those typically reported in infantile Refsum’s disease, namely symmetrical high signal change in periventricular white matter with sparing of subcortical U fibres and pronounced central cerebellar demyelination.5 Involvement of the corpus callosum and thalami may also occur, and on MR spectroscopy decreased N-acetyl-aspartate/creatine and elevated choline/creatine ratios may be found.6 The imaging changes in our patient were not as florid as reported in some childhood cases, perhaps associated with his long survival, but nevertheless contributed to establishing the diagnosis of infantile Refsum’s disease prior to confirmation by genetic testing.
References
1 Waterham HR, Ferdinandusse S, Wanders RJ. Human disorders of peroxisome metabolism and biogenesis. Biochem Biophys Acta 2016; 1863: 922–33.
2 Klouwer FC, Berendse K, Ferdinandusse S et al. Zellweger spectrum disorders: clinical overview and management approach. Orphanet J Rare Dis 2015; 10: 151.
3 Reuber BE, Germain-Lee E, Collins CS et al. Mutations in PEX1 are the most common cause of peroxisome biogenesis disorders. Nat Genet 1997; 17: 445–8.
4 Rosewich H, Ohlenbusch A, Gartner J. Genetic and clinical aspects of Zellweger spectrum patients with PEX1 mutations. J Med Genet 2005; 42: e58.
5 Barth PG, Gootjes J, Bode H et al. Late onset white matter disease in peroxisome biogenesis disorder. Neurology 2001; 57: 1949–55.
6 Kilic M, Karli-Oguz K, Haliloglu G et al. Conventional and advanced MR imaging in infantile Refsum’s disease. Turk J Pediatr 2015; 57: 294–9.
